
Finasteride
An oral anti-androgen API specifically for male pattern hair loss. As a potent 5α-reductase inhibitor, it blocks testosterone conversion to DHT, significantly reducing DHT levels in the scalp and serum.
Overview
Finasteride is an authoritative oral anti-androgen API specifically developed for male pattern hair loss. As a potent 5α-reductase inhibitor, it effectively blocks the conversion of testosterone to dihydrotestosterone (DHT), significantly reducing DHT levels in the scalp and serum. By reversing the follicular miniaturization process, Finasteride promotes hair regrowth and prevents further loss. It is the only oral prescription drug approved by the U.S. FDA for this condition, with a recommended daily dose of 1mg, making it a clinically proven cornerstone in hair growth therapeutics.
Synonyms: finasteride;Finasteridum;Eutiz;Finaszterid;Tealep;Anatine;Avertex;Jacarin;Nasteril;Penester;Pro-Cure;Prostene;Prosterid;Finasterid;Finasterin;Finasterida;Finasteridi;Finasteridas (フィナステリド,非那甾胺;非那司提,MK-906;IVL-3001;MK-0906;YM-152;P-3074); (5alpha,17beta)-(1,1-dimethylethyl)-3-oxo-4-azaandrost-1-ene-17-carboxamide;17beta-(n-tert-butylcarbamoyl)-4-aza-5alpha-androst-1-en-3-one
EINECS: 620-534-3
Product Categories: Hormone Drugs;Biochemistry;Steroids;Steroids (Others);Intermediates & Fine Chemicals;Vitamin Ingredients;Active Pharmaceutical Ingredients;API;Pharmaceuticals;Intracellular receptor;Steroid and Hormone;PROSCAR; Adrenergic alpha-Antagonists
5-alpha Reductase Inhibitors; Drugs Used in Benign Prostatic Hypertrophy; Genitourinary Agents; Hormone Antagonists
Mol File: 98319-26-7.mol
Physicochemical Properties
Melting point: 253 °C
Alpha: 405 -59° (c = 1 in methanol)
Storage temp: room temp
Solubility: DMSO: 32 mg/mL, soluble
Pka: 14.17±0.70(Predicted)
Form: solid
Color: white to beige
Water Solubility: insoluble
MSDS Information
Experimental Data
1.Cell Experiment
Solubility in DMSO: 100 mg/mL (268.43 mM; ultrasonic and warming and heat to 60°C)
Solubility in H2O: < 0.1 mg/mL (ultrasonic) (insoluble)
Preparing Stock Solutions:

Please select an appropriate solvent to prepare a stock solution according to the product's solubility in different solvents. Once prepared, aliquot the solution and store it to avoid product inactivation caused by repeated freeze-thaw cycles.
Stock solution storage and expiration date: -80°C, 2 years; -20°C, 1 year. When stored at -80°C, use within 2 years; when stored at -20°C, use within 1 year.
2.Animal Experiment
Please select an appropriate dissolution protocol based on your experimental animals and route of administration. For the following protocols, first prepare a clear stock solution according to the In Vitro method, and then add the co-solvents sequentially.
To ensure experimental reliability, the clear stock solution can be stored appropriately according to the storage conditions. For in vivo working solutions, it is recommended to prepare them freshly on the day of use.
The percentage indicated before each solvent refers to its volume ratio in the final solution. If precipitation or crystallization occurs during preparation, heating and/or sonication can be used to improve dissolution.
Protocol 1
Add each solvent in the following order:10% DMSO → 90% (20% SBE-β-CD in Saline)
Solubility: ≥ 2.5 mg/mL (6.71 mM); Clear solution
This protocol yields a clear solution of ≥ 2.5 mg/mL (6.71 mM, saturation unknown).
Example for 1 mL of working solution: Take 100 μL of a 25.0 mg/mL clear DMSO stock solution and add it to 900 μL of 20% SBE-β-CD in saline. Mix well.
Protocol 2
Add each solvent in the following order: 10% DMSO → 90% Corn Oil
Solubility: ≥ 2.5 mg/mL (6.71 mM); Clear solution
This protocol yields a clear solution of ≥ 2.5 mg/mL (6.71 mM, saturation unknown). It is not suitable for experiments lasting longer than half a month.
Example for 1 mL of working solution: Take 100 μL of a 25.0 mg/mL clear DMSO stock solution and add it to 900 μL of corn oil. Mix well.
Protocol 3
Add each solvent in the following order: 50% PEG300 → 50% Saline
Solubility: 2 mg/mL (5.37 mM); Suspended solution; Ultrasonication required
Pharmacodynamics
Finasteride is an antiandrogenic compound that works by suppressing the production of serum and intraprostatic dihydrotestosterone (DHT) in men via inhibiting the enzyme responsible for the biosynthesis of DHT. The maximum effect of a rapid reduction in serum DHT concentration is expected to be observed 8 hours following administration of the first dose. In a single man receiving a single oral dose of 5 mg finasteride for up to 4 years, there was a reduction in the serum DHT concentrations by approximately 70% and the median circulating level of testosterone increased by approximately 10-20% within the physiologic range. In a double-blind, placebo-controlled study, finasteride reduced intraprostatic DHT level by 91.4% but finasteride is not expected to decrease the DHT levels to castrate levels since circulating testosterone is also converted to DHT by the type 1 isoenzyme expressed in other tissues. It is expected that DHT levels return to normal within 14 days upon discontinuation of the drug. In a study of male patients with benign prostatic hyperplasia prior to prostatectomy, the treatment with finasteride resulted in an approximate 80% lower DHT content was measured in prostatic tissue removed at surgery compared to placebo. While finasteride reduces the size of the prostate gland by 20%, this may not correlate well with improvement in symptoms. The effects of finasteride are reported to be more pronounced in male patients with enlarged prostates (>25 mL) who are at the greatest risk of disease progression.
In phase III clinical studies, oral administration of finasteride in male patients with male pattern hair loss promoted hair growth and prevented further hair loss by 66% and 83% of the subjects, respectively, which lasted during two years' treatment. The incidences of these effects in treatment groups were significantly higher than that of the group receiving a placebo. Following finasteride administration, the levels of DHT in the scalp skin was shown to be reduced by more than 60%, indicating that the DHT found in scalp is derived from both local DHT production and circulating DHT. The effect of finasteride on scalp DHT is likely seen because of its effect on both local follicular DHT levels as well as serum DHT levels. There is evidence from early clinical observations and controlled studies that finasteride may reduce bleeding of prostatic origin.
Mechanism Of Action
Finasteride acts as a competitive and specific inhibitor of Type II 5α-reductase, a nuclear-bound steroid intracellular enzyme primarily located in the prostatic stromal cell that converts the androgen testosterone into the more active metabolite, 5α-dihydrotestosterone (DHT). DHT is considered to be the primary androgen playing a role in the development and enlargement of the prostate gland. It serves as the hormonal mediator for the hyperplasia upon accumulation within the prostate gland. DHT displays a higher affinity towards androgen receptors in the prostate gland compared to testosterone10 and by acting on the androgen receptors, DHT modulates genes that are responsible for cell proliferation. Responsible for the production of DHT together with type I 5α-reductase, the type II 5α-reductase isozyme is primarily found in the prostate, seminal vesicles, epididymides, and hair follicles as well as liver. Although finasteride is 100-fold more selective for type II 5α-reductase than for the type I isoenzyme, chronic treatment with this drug may have some effect on type I 5α-reductase, which is predominantly expressed in sebaceous glands of most regions of skin, including the scalp, and liver. It is proposed that the type I 5α-reductase and type II 5α-reductase is responsible for the production of one-third and two-thirds of circulating DHT, respectively. The mechanism of action of Finasteride is based on its preferential inhibition of Type II 5α-reductase through the formation of a stable complex with the enzyme in vitro and in vivo. Finasteride works selectively, where it preferentially displays a 100-fold selectivity for the human Type II 5α-reductase over type I enzyme. Inhibition of Type II 5α-reductase blocks the peripheral conversion of testosterone to DHT, resulting in significant decreases in serum and tissue DHT concentrations, minimal to moderate increase in serum testosterone concentrations, and substantial increases in prostatic testosterone concentrations. As DHT appears to be the principal androgen responsible for stimulation of prostatic growth, a decrease in DHT concentrations will result in a decrease in prostatic volume (approximately 20-30% after 6-24 months of continued therapy). It is suggested that increased levels of DHT can lead to potentiated transcription of prostaglandin D2, which promotes the proliferation of prostate cancer cells. In men with androgenic alopecia, the mechanism of action has not been fully determined, but finasteride has shown to decrease scalp DHT concentration to the levels found in the hairy scalp, reduce serum DHT, increase hair regrowth, and slow hair loss. Another study suggests that finasteride may work to reduce bleeding of prostatic origin by inhibiting vascular endothelial growth factor (VEGF) in the prostate, leading to atrophy and programmed cell death. This may bestow the drug therapeutic benefits in patients idiopathic prostatic bleeding, bleeding during anticoagulation, or bleeding after instrumentation.
Similar Products
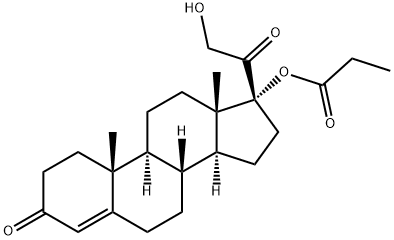
Clascoterone
Therapy
: Dermatology
Target
: AR
CAS
: 19608-29-8
MF
: C24H34O5
MW
: 402.52
Minoxidil
Therapy
: Dermatology
Target
: VGKCs
CAS
: 38304-91-5
MF
: C9H15N5O
MW
: 209.25
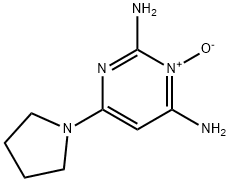
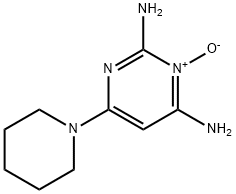
PYRROLIDINYL DIAMINOPYRIMIDINE OXIDE
Therapy
: Dermatology
Target
:
CAS
: 55921-65-8
MF
: C8H13N5O
MW
: 195.22
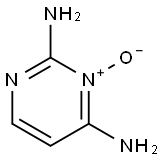
Diaminopyrimidine oxide
Therapy
: Dermatology
Target
:
CAS
: 74638-76-9
MF
: C4H6N4O
MW
: 126.12
