
Thalidomide
An immunomodulator known for its teratogenicity; currently used to treat erythema nodosum leprosy and multiple myeloma.
概况
Thalidomide is an effective oral immunomodulatory drug and anti-angiogenic agent. It exerts its pharmacological effects by regulating the expression of key cytokines such as tumor necrosis factor-alpha (TNF-α), inhibiting abnormal angiogenesis, and modulating the function of immune cells (e.g., T cells, B cells). Thalidomide can be used in the research of multiple myeloma, leprosy (Hansen's disease)-associated erythema nodosum leprosum, certain autoimmune diseases (e.g., systemic lupus erythematosus), and specific solid tumors (e.g., liver cancer, gastric cancer).
Synonyms: NSC-66847; NSC-527179; K-17; FPF-300; FPF300; Talizer; Thalidomide Celgene; Thalidomide Pharmion; Synovir; Thalomid; Thaled; (+)-N-(2,6-dioxo-3-Piperidyl)phthalimide; (+)-Thalidomide; (±)-N-(2,6-dioxo-3-piperidyl)phthalimide; (±)-thalidomide; 1,3-dioxo-2-(2,6-dioxopiperidin-3-yl)isoindoline; 2,6-dioxo-3-phthalimidopiperidine; 3-Phthalimidoglutarimide; alpha-(N-Phthalimido)glutarimide; alpha-N-Phthalylglutaramide; N-(2,6-dioxo-3-piperidyl)phthalimide; N-Phthaloylglutamimide; N-Phthalyl-glutaminsaeure-imid; N-Phthalylglutamic acid imide; Talidomida; Thalidomide; Thalidomidum; α-(N-phthalimido)glutarimide; α-N-phthalylglutaramide; α-phthalimidoglutarimide
EINECS: 200-031-1
Product Categories: Intermediates & Fine Chemicals; Pharmaceuticals; API; Cytokine signaling; Influenza viruses; Angiogenesis and metastasis; Inhibitor; Antibiotics
Mol File: 50-35-1.mol
物化性质
Melting point: 269-271°C
Storage temp: 2-8℃
Solubility: 45% (w/v) aq 2-hydroxypropyl-β-cyclodextrin: 0.6 mg/mL
Form: Powder
Color: White to Off-white
Stability: Flammable; Incompatible with strong oxidizing agents.
MSDS信息
实验数据
1. Cell Experiment
Solubility in DMSO: 100 mg/mL (387.25 mM; solubilized by ultrasound; hygroscopic DMSO significantly affects product solubility, so use freshly opened DMSO).
Preparing Stock Solutions:

Please prepare the stock solution in the appropriate solvent based on the product's solubility in different solvents. Once prepared, aliquot and store to avoid product degradation caused by repeated freeze-thaw cycles.
Stock solution storage and expiration date: -80°C, 1 year; -20°C, 6 months. When stored at -80°C, use within 1 year; when stored at -20°C, use within 6 months.
2. Animal Experiment
Please select the appropriate dissolution method based on your experimental animals and administration method.
For the following dissolution methods, first prepare a clear stock solution in vitro, then add cosolvents in sequence:
To ensure reliable experimental results, the clear stock solution can be stored appropriately according to storage conditions. For in vivo experiments, it is recommended that the working solution be prepared fresh and used the same day.
The percentages shown before the solvents below refer to the volume percentage of the solvent in the final solution. If precipitation or precipitation occurs during the preparation process, heating and/or sonication can be used to aid dissolution.
Protocol 1
Add each solvent in order: 10% DMSO, 40% PEG300, 5% Tween-80, 45% Saline.
Solubility: ≥ 2.5 mg/mL (9.68 mM); Clear Solution
This protocol produces a clear solution with a concentration of ≥ 2.5 mg/mL (saturation unknown).
For a 1 mL working solution, add 100 μL of the 25.0 mg/mL clear DMSO stock solution to 400 μL of PEG300 and mix thoroughly. Add 50 μL of Tween-80 to the solution and mix thoroughly. Then, add 450 μL of normal saline to bring the volume to 1 mL.
To prepare normal saline: Dissolve 0.9 g of sodium chloride in ddH₂O and dilute to 100 mL. This will yield a clear, transparent normal saline solution.
Protocol 2
Add each solvent in order: 10% DMSO, 90% corn oil.
Solubility: ≥ 2.5 mg/mL (9.68 mM); Clear solution
This protocol produces a clear solution of ≥ 2.5 mg/mL (saturation unknown). This protocol should be used for animal experiments with experimental durations exceeding two weeks.
For a 1 mL working solution, add 100 μL of the 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix thoroughly.
For the following dissolution schemes, prepare the working solution directly. It is recommended to prepare immediately and use it as soon as possible. The percentages shown before the solvents below refer to the volume percentage of the final solution. If precipitation or precipitation occurs during the preparation process, heating and/or sonication can be used to aid dissolution.
Protocol 1
Add each solvent in sequence: 0.5% CMC-Na/saline water
Solubility: 20 mg/mL (77.45 mM); Suspension; Ultrasonication
Protocol 2
Add each solvent in sequence: 10% Tween-80 in PBS
Solubility: 20 mg/mL (77.45 mM); Suspension; Ultrasonication
Pharmacodynamics
Thalidomide, originally developed as a sedative, is an immunomodulatory and anti-inflammatory agent with a spectrum of activity that is not fully characterized. However, thalidomide is believed to exert its effect through inhibiting and modulating the level of various inflammatory mediators, particularly tumor necrosis factor-alpha (TNF-a) and IL-6. Additionally, thalidomide is also shown to inhibit basic fibroblast growth factor (bFGF) and vascular endothelial growth factor (VEGF), suggesting a potential anti-angiogenic application of thalidomide in cancer patients.
Thalidomide is racemic — it contains both left and right handed isomers in equal amounts: the (+)R enantiomer is effective against morning sickness, and the (−)S enantiomer is teratogenic. The enantiomers are interconverted to each other in vivo; hence, administering only one enantiomer will not prevent the teratogenic effect in humans.
Mechanism Of Action
The mechanism of action of thalidomide is not fully understood. Previous research indicate that thalidomide binds to cerebron, a component of the E3 ubiquitin ligase complex, to selectively degrade the transcription factor IKZF3 and IKZF1. These 2 transcription factors are vital for the proliferation and survival of malignant myeloma cells. Regarding TNF-alpha, thalidomide seems to block this mediator via a variety of mechanism. Thalidomide can inhibit the expression myeloid differentiating factor 88 (MyD88), an adaptor protein that is involved in the TNF-alpha production signalling pathway, at the protein and RNA level. Additionally, thalidomide prevents the activation of Nuclear Factor Kappa B (NF-kB), another upstream effector of the TNF-alpha production pathway. Finally, some evidences suggest that thalidomide can block alpha-1 acid glycoprotein (AGP), a known inducer of the NF-kB/MyD88 pathway, thus inhibiting the expression of TNF-alpha. The down-regulation of NF-kB and MyD88 can also affect the cross talk between the NF-kB/MyD88 and VEGF pathway, resulting in thalidomide's anti-angiogenic effect.
相似产品
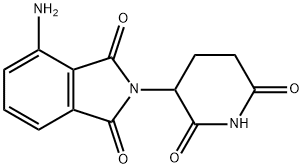
Pomalidomide
治疗领域
: Oncology
作用靶点
: TNFα;IKZF1;IKZF3;IL6
CAS
: 19171-19-8
分子式
: C13H11N3O4
分子量
: 273.24
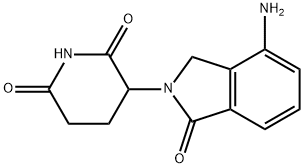
Lenalidomide
治疗领域
: Oncology
作用靶点
: CSNK1A1;IKZF1;IKZF3;CRBN
CAS
: 191732-72-6
分子式
: C13H13N3O3
分子量
: 259.261
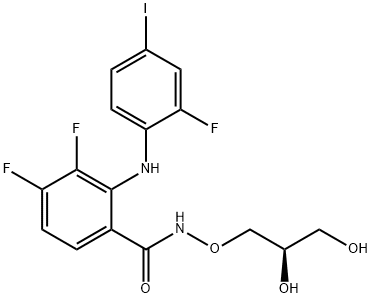
Mirdametinib
治疗领域
: Oncology
作用靶点
: MEK2; MEK1
CAS
: 391210-10-9
分子式
: C16H14F3IN2O4
分子量
: 482.19
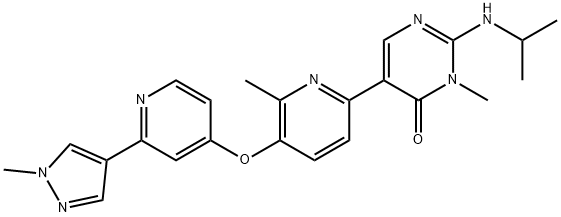
Vimseltinib
治疗领域
: Oncology
作用靶点
: CSF1R
CAS
: 1628606-05-2
分子式
: C23H25N7O2
分子量
: 431.49
