
Lenalidomide
An analog of thalidomide; an immunomodulatory agent used to treat multiple myeloma and myelodysplastic syndrome.
Overview
Lenalidomide is a potent oral immunomodulatory drug (IMiD) that exerts immunomodulatory, anti-angiogenic, and direct anti-tumor cell proliferation effects by binding to the cereblon (CRBN) protein and regulating the ubiquitin-proteasome system. Lenalidomide can be used in the research of hematological malignancies such as multiple myeloma (MM), myelodysplastic syndromes (MDS, especially those with 5q deletion), and mantle cell lymphoma (MCL), as well as scientific research on related immunomodulatory mechanisms and tumorigenesis and development.
Synonyms: IMID-5013; CDC-5013; CDC-501; CC-5013; IMiD-3; ENMD-0997; Revimid (former Brand Name); Leavdo; Zelvina; Lenalidomidum; Lenalidomida; Ladevina; 1-oxo-2-(2,6-dioxopiperidin-3-yl)-4-aminoisoindoline; 3-(4-Amino-1-oxoisoindolin-2-yl)piperidine-2,6-dione; Lena
EINECS: 691-297-1
Product Categories: Molecular targeted antineoplastic; Intermediates & Fine Chemicals; Pharmaceuticals; Amines; Heterocycles; CC-5013; Inhibitors; Pharmaceutical intermediate; API
Mol File: 191732-72-6.mol
Physicochemical Properties
Melting point: 265-268℃
Storage temp: 2-8°C
Solubility: Dissolved in DMSO
Form: Solid
Color: Off-white to Light Yellow
Stability: Solutions in DMSO can be stored at -20 °C for up to 3 months.
MSDS Information
Experimental Data
1. Cell Experiment
Solubility in DMSO: 100 mg/mL (385.71 mM; solubilized by ultrasound; hygroscopic DMSO significantly affects product solubility, so use freshly opened DMSO).
Preparing Stock Solutions:
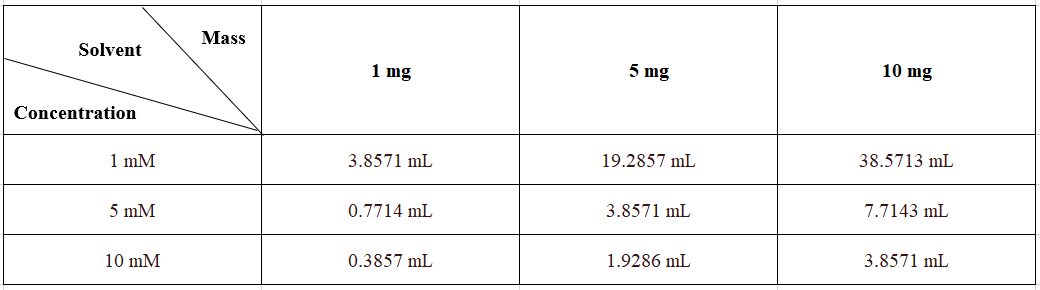
Please prepare the stock solution in the appropriate solvent based on the product's solubility in different solvents. Once prepared, aliquot and store to avoid product degradation caused by repeated freeze-thaw cycles.
Storage and expiration date of the stock solution: -80°C, 1 year; -20°C, 6 months. When stored at -80°C, use within 1 year; when stored at -20°C, use within 6 months.
2. Animal Experiment
Please select the appropriate dissolution method based on your experimental animal and administration method.
For the following dissolution methods, first prepare a clear stock solution in vitro, then add cosolvents in sequence:
To ensure reliable experimental results, the clear stock solution can be stored appropriately according to storage conditions. For in vivo experiments, it is recommended that you prepare the working solution immediately and use it the same day.
The percentage displayed before the solvent below refers to the volume percentage of the solvent in the final solution. If precipitation or precipitation occurs during preparation, heating and/or sonication can be used to aid dissolution.
Protocol 1
Add each solvent in order: 10% DMSO, 40% PEG300, 5% Tween-80, 45% Saline.
Solubility: ≥ 2.5 mg/mL (9.64 mM); Clear Solution
This protocol produces a clear solution with a concentration of ≥ 2.5 mg/mL (saturation unknown).
For a 1 mL working solution, add 100 μL of the 25.0 mg/mL clear DMSO stock solution to 400 μL of PEG300 and mix thoroughly. Add 50 μL of Tween-80 to the solution and mix thoroughly. Then, add 450 μL of normal saline to bring the volume to 1 mL.
To prepare normal saline: Dissolve 0.9 g of sodium chloride in ddH₂O and dilute to 100 mL. This will yield a clear, transparent normal saline solution.
Protocol 2
Add each solvent in sequence: 10% DMSO, 90% (20% SBE-β-CD in saline).
Solubility: ≥ 2.5 mg/mL (9.64 mM); Clear Solution
This option yields a clear solution of ≥ 2.5 mg/mL (saturation unknown).
For a 1 mL working solution, add 100 μL of the 25.0 mg/mL clear DMSO stock solution to 900 μL of a 20% SBE-β-CD solution in saline and mix thoroughly.
Add 2 g of SBE-β-CD (sulfobutyl ether β-cyclodextrin) powder to 10 mL of saline and dissolve completely until clear.
Protocol 3
Add each solvent in sequence: 10% DMSO, 90% corn oil.
Solubility: ≥ 2.5 mg/mL (9.64 mM); Clear solution
This protocol produces a clear solution of ≥ 2.5 mg/mL (saturation unknown). This protocol should be used for animal experiments with experimental durations exceeding two weeks.
For a 1 mL working solution, add 100 μL of the 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix thoroughly.
Pharmacodynamics
In hematological malignancies, the immune system is deregulated in the form of altered cytokine networks in the tumour microenvironment, defective T cell regulation of host-tumour immune interactions, and diminished NK cell activity. Lenalidomide is an immunomodulatory agent with antineoplastic, antiangiogenic, and anti-inflammatory properties. Lenalidomide exerts direct cytotoxicity by increasing apoptosis and inhibiting the proliferation of hematopoietic malignant cells. It delays tumour growth in nonclinical hematopoietic tumour models in vivo, including multiple myeloma. Lenalidomide also works to limit the invasion or metastasis of tumour cells and inhibits angiogenesis.
Lenalidomide also mediates indirect antitumour effects via its immunomodulatory actions: it inhibits the production of pro-inflammatory cytokines, which are implicated in various hematologic malignancies. Lenalidomide enhances the host immunity by stimulating T cell proliferation and enhancing the activity of natural killer (NK) cells. Lenalidomide is about 100–1000 times more potent in stimulating T cell proliferation than thalidomide. In vitro, it enhances antibody-dependent cell-mediated cytotoxicity (ADCC), which is even more pronounced when used in combination with rituximab. Due to its anti-inflammatory properties, lenalidomide has been investigated in the context of inflammatory and autoimmune diseases, such as amyotrophic lateral sclerosis.
Mechanism Of Action
Lenalidomide is a drug with multiple mechanisms of action. Lenalidomide exerts immunomodulating effects by altering cytokine production, regulating T cell co-stimulation, and enhancing the NK cell-mediated cytotoxicity. Lenalidomide directly inhibits the cullin ring E3 ubiquitin ligase complex: upon binding to cereblon, a substrate adaptor of the complex, lenalidomide modulates substrate specificity of the complex to recruit substrate proteins of the ligase, including Ikaros (IKZF1), Aiolos (IKZF3), and CK1α. These substrates are then tagged for ubiquitination and subsequent proteasomal degradation. IKZF1 and IKZF3 are B-cell transcription factors that are essential for B-cell differentiation and survival of malignant cells. IKZF3 also regulates the expression of interferon regulatory factor 4 (IRF4), which is a transcription factor that regulates the aberrant myeloma-specific gene. The immunomodulatory actions of lenalidomide can be partly explained by the degradation of IKZF3, since it is a repressor of the interleukin 2 gene (IL2): as lenalidomide decreases the level of IKZF3, the production of IL-2 increases, thereby increasing the proliferation of natural killer (NK), NKT cells, and CD4+ T cells. Lenalidomide inhibits the production of pro-inflammatory cytokines TNF-α, IL-1, IL-6, and IL-12, while elevating the production of anti-inflammatory cytokine IL-10. Lenalidomide acts as a T-cell co-stimulatory molecule that promotes CD3 T-cell proliferation and increases the production of IL-2 and IFN-γ in T lymphocytes, which enhances NK cell cytotoxicity and ADCC. It inhibits the expression and function of T-regulatory cells, which are often overabundant in some hematological malignancies. Lenalidomide directly exerts antitumour effects by inhibiting the proliferation and inducing apoptosis of tumour cells. Lenalidomide triggers the activation of pro-apoptotic caspase-8, enhances tumour cell sensitivity to FAS-induced apoptosis, and downregulates NF-κB, an anti-apoptotic protein. Independent of its immunomodulatory effects, lenalidomide mediates anti-angiogenic effects by inhibiting angiogenic growth factors released by tumour cells, such as vascular endothelial growth factor (VEGF), basic fibroblastic-growth factor (BFGF), and hepatocyte-growth factor. In vitro, lenalidomide inhibits cell adhesion molecules such as ICAM-1, LFA-1, β2 and β3 integrins, as well as gap-junction function, thereby preventing metastasis of malignant cells.
Similar Products
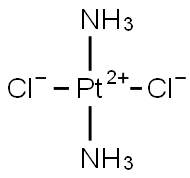
Cisplatin
Therapy
: Oncology
Target
: DNA
CAS
: 15663-27-1
MF
: Cl2H6N2Pt
MW
: 300.04
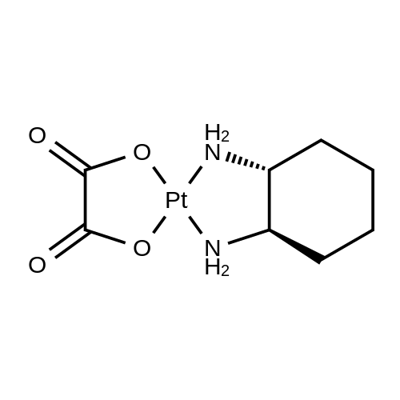
Oxaliplatin
Therapy
: Oncology
Target
: DNA
CAS
: 61825-94-3
MF
: C8H12N2O4Pt
MW
: 395.28
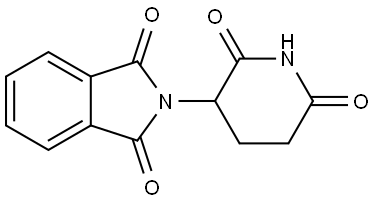
Thalidomide
Therapy
: Oncology
Target
: Ligands for E3 Ligase; Autophagy; Apoptosis; Molecular Glues
CAS
: 50-35-1
MF
: C13H10N2O4
MW
: 258.23
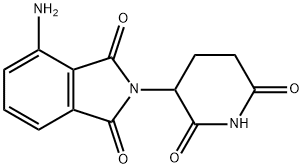
Pomalidomide
Therapy
: Oncology
Target
: TNFα;IKZF1;IKZF3;IL6
CAS
: 19171-19-8
MF
: C13H11N3O4
MW
: 273.24
