
Clomiphene Citrate
A selective estrogen receptor modulator (SERM); mainly used to induce ovulation and treat female infertility.
Overview
Clomiphene Citrate is a selective estrogen receptor modulator (SERM) that exerts both anti-estrogenic and weak estrogenic activity. It is used in research related to gynecological reproductive dysfunction, such as anovulatory infertility and polycystic ovary syndrome, as well as oligospermia and hypogonadism in men.
Synonyms: NSC-35770; MER-41; MRL-41; Clomid; Serophene; Clomifene citrate; Clomifenum; Omifin; Clomiphane citrate; Chloramiphene; Clomifene dihydrogen citrate; Clomifeno; Clomiphene dihydrogen citrate; Clomiphene-r; Clomiphine; Clomivid; Clostilbegyt; 2-[4-(2-chloro-1,2-diphenylethenyl)-phenoxy]-n,n-diethylethanamine; Clomiphene citrate salt; Clomphid; Pergotime; 1-(p-(beta-diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylenecitrate; 2-(p-(2-chloro-1,2-diphenylvinyl)phenoxy)-triethylamincitrate(1:1); 2-chloro-1-(p-(beta-diethylaminoethoxy)phenyl)-1,2-diphenylethylenecitrate
EINECS: 200-035-3
Product Categories: API; Intermediates & Fine Chemicals; Pharmaceuticals; Amines; Aromatics; Peptides; Pharmaceutical intermediates; Hormone regulation
Mol File: 50-41-9.mol
Physicochemical Properties
Melting point: 116.5-118°C
Storage temp: 2-8°C
Solubility: Slightly soluble in water, methanol, chloroform, and DMSO. Insoluble in ether.
Form: Powder
Color: White to Off-white
Stability: Hygroscopicity; Light Sensitive
MSDS Information
Experimental Data
1. Cell Experiment
Solubility in DMSO: ≥ 50 mg/mL (83.60 mM; Hygroscopic DMSO significantly affects product solubility, so use freshly opened DMSO.)
Solubility in H2O: 1 mg/mL (1.67 mM; solubilized with ultrasound)
* "≥" means soluble, but saturation unknown.
Preparing Stock Solutions:
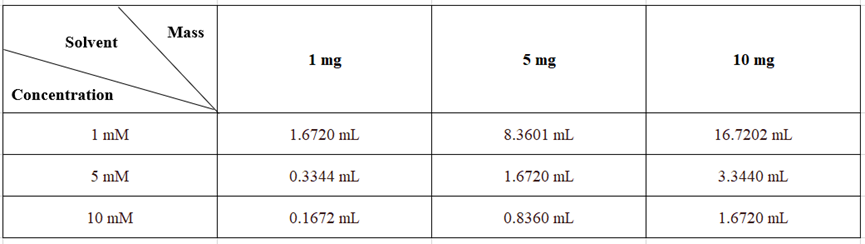
Please prepare the stock solution based on the product's solubility in different solvents. Once prepared, aliquot and store to avoid product degradation caused by repeated freeze-thaw cycles.
Stock solution storage and expiration date: -80°C, 6 months; -20°C, 1 month (sealed, moisture-free). When stored at -80°C, use within 6 months; when stored at -20°C, use within 1 month.
Note: If you choose water as the stock solution, please dilute it to the working solution and then filter it through a 0.22 μm filter membrane for sterilization before use.
2. Animal Experiment
Please select the appropriate dissolution method based on your experimental animal and administration method.
For the following dissolution methods, first prepare a clear stock solution in vitro, then add cosolvents in sequence:
To ensure reliable experimental results, the clear stock solution can be stored appropriately according to storage conditions. For in vivo experiments, it is recommended that you prepare the working solution immediately and use it the same day.
The percentage displayed before the solvent below refers to the volume percentage of the solvent in the final solution. If precipitation or precipitation occurs during preparation, heating and/or sonication can be used to aid dissolution.
Protocol 1
Add each solvent in order: 10% DMSO, 40% PEG300, 5% Tween-80, 45% Saline.
Solubility: ≥ 2.5 mg/mL (4.18 mM); Clear Solution
This protocol produces a clear solution with a concentration of ≥ 2.5 mg/mL (saturation unknown).
For a 1 mL working solution, add 100 μL of the 25.0 mg/mL clear DMSO stock solution to 400 μL of PEG300 and mix thoroughly. Add 50 μL of Tween-80 to the solution and mix thoroughly. Then, add 450 μL of normal saline to bring the volume to 1 mL.
To prepare normal saline, dissolve 0.9 g of sodium chloride in ddH₂O and dilute to 100 mL. This will yield a clear, transparent normal saline solution.
Protocol 2
Add each solvent one by one: 10% DMSO, 90% (20% SBE-β-CD in Saline)
Solubility: ≥ 2.5 mg/mL (4.18 mM); Clear solution
This protocol yields a clear solution of ≥ 2.5 mg/mL (saturation unknown).
Taking 1 mL working solution as an example, add 100 μL DMSO stock solution (25.0 mg/mL) to 900 μL 20% SBE-β-CD in Saline, and mix evenly.
Preparation of 20% SBE-β-CD in Saline (4°C, storage for one week): 2 g SBE-β-CD powder is dissolved in 10 mL Saline, completely dissolve until clear.
Protocol 3
Add each solvent one by one: 10% DMSO, 90% Corn Oil
Solubility: ≥ 2.5 mg/mL (4.18 mM); Clear solution
This protocol yields a clear solution of ≥ 2.5 mg/mL (saturation unknown). If the continuous dosing period exceeds half a month, please choose this protocol carefully.
Taking 1 mL working solution as an example, add 100 μL DMSO stock solution (25.0 mg/mL) to 900 μL Corn oil, and mix evenly.
For the following dissolution methods, please prepare the working solution directly. It is recommended to prepare fresh solutions and use them promptly within a short period of time.
The percentages shown for the solvents indicate their volumetric ratio in the final prepared solution. If precipitation or phase separation occurs during preparation, heat and/or sonication can be used to aid dissolution.
Protocol 1
Add each solvent one by one: PBS
Solubility: 7.14 mg/mL (11.94 mM); Clear solution; Need ultrasonic and warming and heat to 60°C
Pharmacodynamics
Clomifene (previously clomiphene) is an orally administered, non steroidal, ovulatory stimulant that acts as a selective estrogen receptor modulator (SERM). Clomifene can lead to multiple ovulation, and hence increase the risk of conceiving twins. In comparison to purified FSH, the rate of ovarian hyperstimulation syndrome is low. There may be an increased risk of ovarian cancer and weight gain. Clomifene is capable of interacting with estrogen-receptor-containing tissues, including the hypothalamus, pituitary, ovary, endometrium, vagina, and cervix. It may compete with estrogen for estrogen-receptor-binding sites and may delay replenishment of intracellular estrogen receptors. Clomifene initiates a series of endocrine events culminating in a preovulatory gonadotropin surge and subsequent follicular rupture. The first endocrine event, in response to a course of clomifene therapy, is an increase in the release of pituitary gonadotropins. This initiates steroidogenesis and folliculogenesis resulting in growth of the ovarian follicle and an increase in the circulating level of estradiol. Following ovulation, plasma progesterone and estradiol rise and fall as they would in a normal ovulatory cycle.
Mechanism Of Action
Clomifene has both estrogenic and anti-estrogenic properties, but its precise mechanism of action has not been determined. Clomifene appears to stumulate the release of gonadotropins, follicle-stimulating hormone (FSH), and leuteinizing hormone (LH), which leads to the development and maturation of ovarian follicle, ovulation, and subsequent development and function of the coprus luteum, thus resulting in pregnancy. Gonadotropin release may result from direct stimulation of the hypothalamic-pituitary axis or from a decreased inhibitory influence of estrogens on the hypothalamic-pituitary axis by competing with the endogenous estrogens of the uterus, pituitary, or hypothalamus. Clomifene has no apparent progestational, androgenic, or antrandrogenic effects and does not appear to interfere with pituitary-adrenal or pituitary-thyroid function.
Similar Products
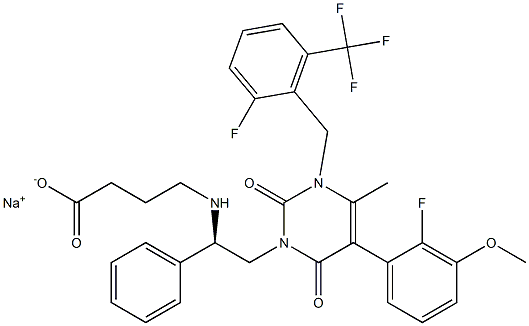
Elagolix Sodium
Therapy
: Women's health
Target
: GnRHR
CAS
: 832720-36-2
MF
: C32H29F5N3O5.Na
MW
: 653.572
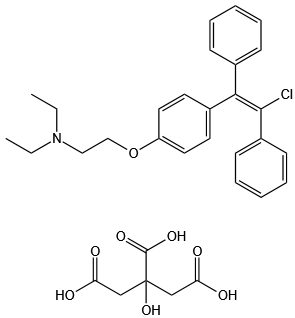
trans-Clomiphene Citrate
Therapy
: Women's health
Target
: Estrogen Receptor/ERR
CAS
: 7599-79-3
MF
: C32H36ClNO8
MW
: 598.08314
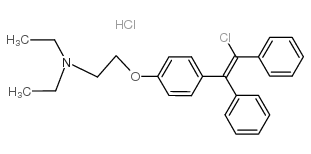
CIS-CLOMIPHENE HCL
Therapy
: Genitourinary
Target
: ERs
CAS
: 15690-55-8
MF
: C26H29Cl2NO
MW
: 442.421
