
Probenecid
A urate transporter inhibitor, a uricosuric agent. It is used in the treatment of gouty arthritis, tophaceous gout, and hyperuricemia.
Overview
Probenecid, the prototypical uricosuric agent, is an organic anion transporter inhibitor. It reduces tubular reabsorption of urate to promote uric acid excretion while inhibiting renal excretion of organic anions, making it suitable as an adjunct to antibacterial therapy.
Synonyms: TD-213; Benuryl; Benemid; PanCytoVir; Probenecidum; 4-((dipropylamino)sulfonyl)-benzoicaci; 4-(Dipropylsulfamoyl)benzoicacid; 4-(dipropylsulfamoyl)benzoicaciChemicalbookd; 4-[(dipropylamino)sulfonyl]-benzoicaci; Apurina; benacen; Benemid; 4-[(dipropylamino)sulphonyl]benzoicacid
EINECS: 200-344-3
Product Categories: Miscellaneous; API; Intermediates&FineChemicals; Pharmaceuticals; Sulfur&SeleniumCompounds; Vanilloid/TRPVchannel; chemicaladditive; phChemicalbookarmaceuticalintermediate; PROBENEMID; pharmaceuticalchemicals; Pharmaceuticalrawmaterials; Adjuvants, Pharmaceutic; Amides; Antigout Preparations; Antirheumatic Agents; Cytochrome P-450 CYP2C19 Inhibitors; Cytochrome P-450 CYP2C8 Inducers; Drugs that are Mainly Renally Excreted; Genito Urinary System and Sex Hormones; Musculo-Skeletal System
Mol File: 57-66-9.mol
Physicochemical Properties
Melting point: 194-196°C
Storage temp: Store at RT
Solubility: DMSO (Slightly), Methanol (Slightly)
Form: Solid
Color: White to Off-White
Water Solubility: <0.1 g/100 mL at 20 ºC
Stability: Stable, but may be light sensitive. Incompatible with strong oxidizing agents.
MSDS Information
Experimental Data
1. Cell Experiment
DMSO : ≥ 100 mg/mL (350.43 mM)
0.5 M NaOH : 100 mg/mL (350.43 mM; ultrasonic and adjust pH to 12 with NaOH)
* "≥" means soluble, but saturation unknown.
Preparing Stock Solutions:

Please select an appropriate solvent for preparing stock solutions based on the product's solubility in different solvents. Once prepared, aliquot and store the solution to avoid product degradation caused by repeated freeze-thaw cycles.
Storage conditions and shelf life for stock solutions: -80°C, 1 year; -20°C, 6 months.
When stored at -80°C, use within 1 year; when stored at -20°C, use within 6 months.
2. Animal Experiment
Please select an appropriate dissolution protocol based on your experimental animals and route of administration. For the following protocols, first prepare a clear stock solution following the in vitro method, then add cosolvents sequentially:
To ensure experimental reliability, the clear stock solution can be appropriately stored according to storage conditions. For in vivo working solutions, it is recommended to prepare them fresh and use on the same day. The percentage indicated before each solvent represents its volume ratio in the final solution. If precipitation occurs during preparation, heating and/or ultrasonication can be used to aid dissolution.
Protocol 1
Add solvent in order: 5% Sodium bicarbonate in ddH₂O
Solubility: 16 mg/mL (56.07 mM); Clear solution; Ultrasonication required
Protocol 2
Add solvents in order: 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% saline
Solubility: 2.5 mg/mL (8.76 mM); Suspended solution; Ultrasonication required
This protocol yields a homogeneous suspension of 2.5 mg/mL (8.76 mM), which can be used for oral and intraperitoneal administration.
Example for 1 mL working solution: Add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix well. Add 50 μL Tween-80 to the mixture and mix thoroughly. Then add 450 μL saline to bring the volume to 1 mL.
Protocol 3
Add solvents in order: 10% DMSO → 90% corn oil
Solubility: ≥ 2.5 mg/mL (8.76 mM); Clear solution
This protocol yields a clear solution of ≥ 2.5 mg/mL (8.76 mM, saturation unknown). This method is not suitable for experiments lasting longer than half a month.
Example for 1 mL working solution: Add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL corn oil and mix well.
Pharmacodynamics
Probenecid is a uricosuric and renal tubular blocking agent and is used in combination with colchicine to treat chronic gouty arthritis when complicated by frequent, recurrent acute attacks of gout. It inhibits the reabsorption of urate at the proximal convoluted tubule, thus increasing the urinary excretion of uric acid and decreasing serum urate levels. Effective uricosuria reduces the miscible urate pool, retards urate deposition, and promotes resorption of urate deposits. At the proximal and distal tubles, probenecid competitively inhibits the secretion of many weak organic acids including penicillins, most cephalosporins, and some other β-lactam antibiotics. This results in an increase in the plasma concentrations of acidic drugs eliminated principally by renal secretion, but only a slight increase if the drug is eliminated mainly by filtration. Thus, the drug can be used for therapeutic advantages to increase concentrations of certain β-lactam antibiotics in the treatment of gonorrhea, neurosyphilis, or pelvic inflammatory disease (PID).
Mechanism Of Action
Probenecid inhibits the tubular reabsorption of urate, thus increasing the urinary excretion of uric acid and decreasing serum urate levels. Probenecid may also reduce plasma binding of urate and inhibit renal secretion of uric acid at subtherapeutic concentrations. The mechanism by which probenecid inhibits renal tubular transport is not known, but the drug may inhibit transport enzymes that require a source of high energy phosphate bonds and/or nonspecifically interfere with substrate access to protein receptor sites on the kidney tubules.
Similar Products
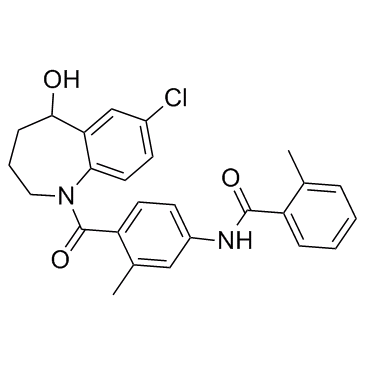
Tolvaptan
Therapy
: Genitourinary
Target
: V2R
CAS
: 150683-30-0
MF
: C26H25ClN2O3
MW
: 448.94
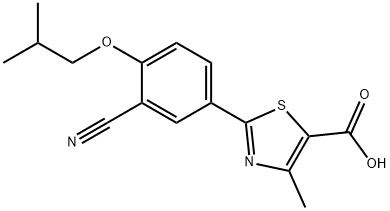
Febuxostat
Therapy
: Musculoskeletal diseases
Target
: XO
CAS
: 144060-53-7
MF
: C16H16N2O3S
MW
: 316.37
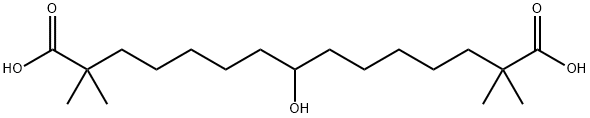
Bempedoic acid
Therapy
: Cardiometabolic diseases
Target
: ACLY
CAS
: 738606-46-7
MF
: C19H36O5
MW
: 344.49
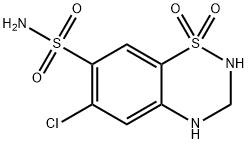
Hydrochlorothiazide
Therapy
: Cardiometabolic diseases
Target
: Thiazide-sensitive sodium-chloride cotransporter
CAS
: 58-93-5
MF
: C7H8ClN3O4S2
MW
: 297.74
