Review of the Development History of TACs Multifunctional Linkers
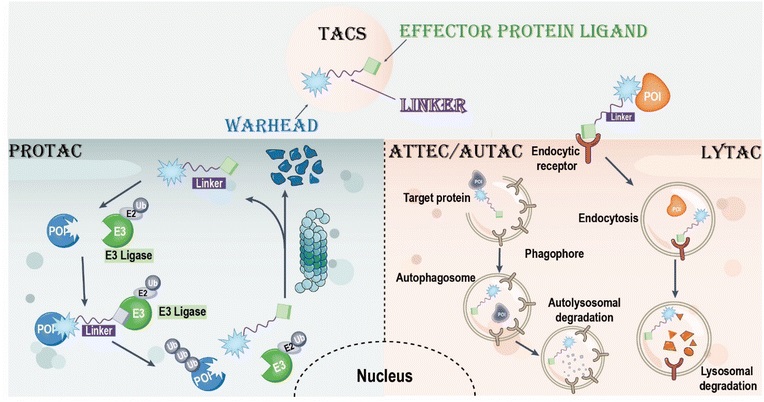
Targeted chimeras (TACs, such as PROTACs and LYTACs) represent a potential strategy for the selective degradation of difficult-to-drug proteins, acting through bifunctional molecules or molecular glues to provide novel therapies for diseases that are difficult to treat with traditional small molecules. As a bridge connecting target protein ligands and effector protein ligands, the linker of TACs is crucial to molecular conformation and activity. Recent advancements in photo-controlled and cleavable linker design have improved the efficiency and spatiotemporal control of TACs, but challenges remain, such as optimizing stability.
Traditional small molecule inhibitors are ineffective against "undruggable" targets (such as proteins without active pockets) and are prone to drug resistance due to target protein mutations. This predicament has long constrained the progress of drug development. However, targeted chimeras (TACs), with their precise regulation of linkers, can not only efficiently degrade target proteins but also overcome drug resistance problems, becoming a core force in moving targeted protein degradation (TPD) technology from proof of concept to clinical application.
1. Traditional Linkers
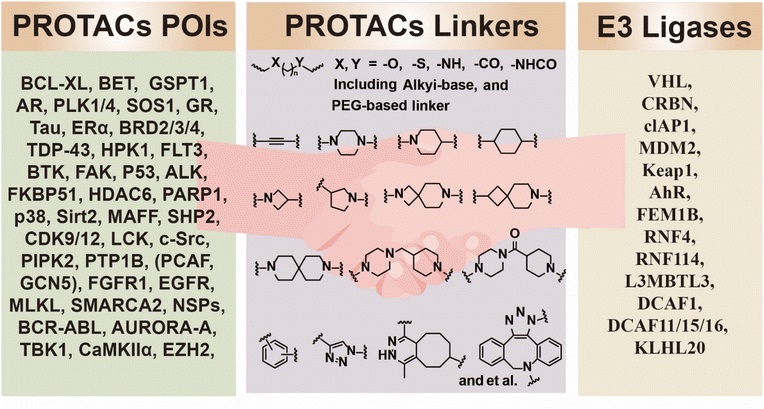
The typical structure of TACs consists of three parts: target protein ligand (binding pathogenic protein) + effector protein ligand (such as E3 ubiquitin ligase ligand, lysosomal receptor ligand) + linker. The physicochemical properties of the linker directly determine the spatial conformation, ternary complex stability, and pharmacokinetic performance of the TACs. Traditional linkers are designed with chemical structure as the core and are mainly divided into the following two categories.
1.1 Singleline Linkers
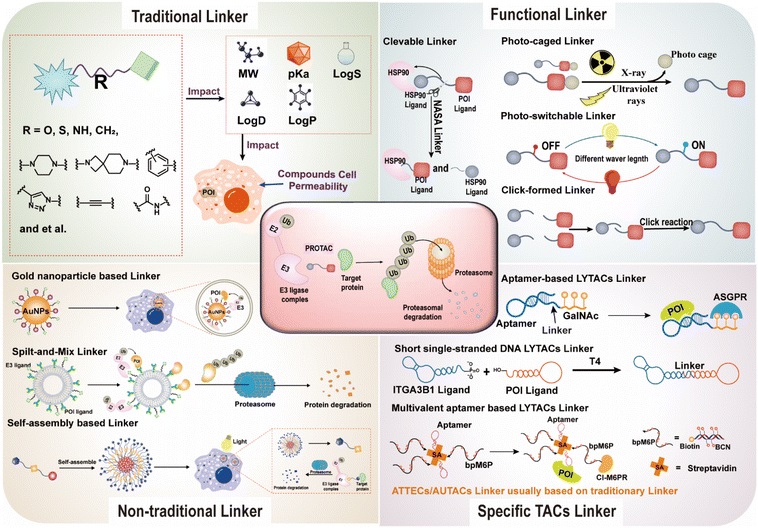
Based on their conformational properties, singlet linkers can be further divided into flexible and rigid types, adapting to the binding requirements of different target proteins and effector proteins. Flexible linkers, including PEG-based, alkyl-based, and amide-based types, can flexibly adapt to different spatial distances between target proteins (POIs) and effector proteins through conformational adjustments. A typical example is the PEG-based linker—which significantly improves the water solubility and cell permeability of PROTACs degrading BRD2/3/4. Rigid linkers, including cycloalkane-based, spirocyclic, and aryl (such as triazole) types, can fix the molecular conformation of TACs, reduce non-specific binding, and improve targeting precision.
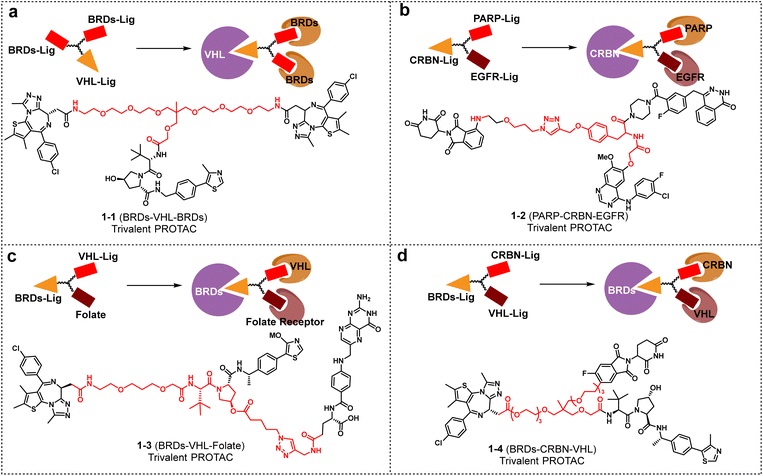
1.2 Y-type trivalent linker
Overcoming the limitations of traditional bivalent design, the Y-type trivalent linker connects multiple ligands through a branched core, significantly improving binding valence and degradation efficiency.
2. Functionalized Linkers
To reduce the off-target toxicity of TACs, functionalized linkers introduce responsive groups such as light, enzymes, and chemical signals to achieve "on-demand regulation" of degradation activity, providing possibilities for precision therapy.
2.1 Click Chemistry Linkers
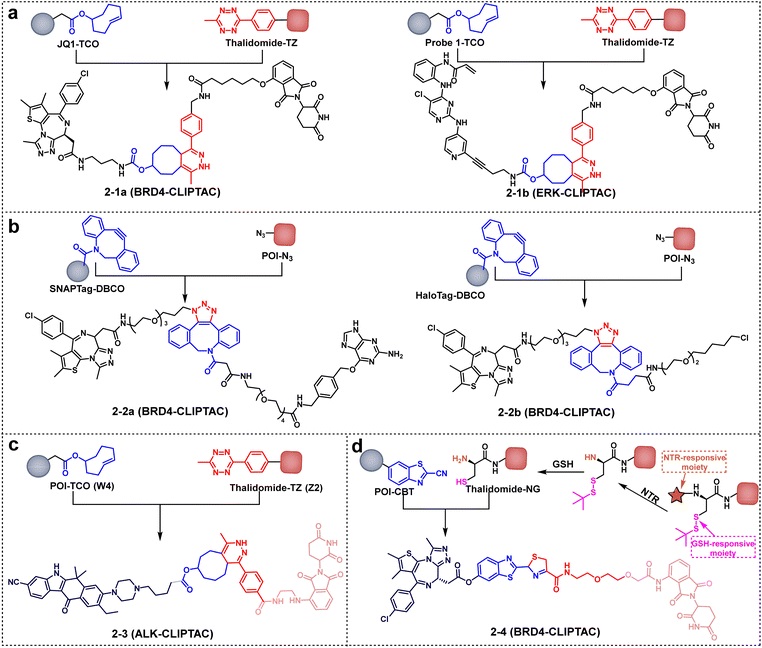
TACs are split into two low-molecular-weight fragments: fragment 1 (target protein ligand + TCO) and fragment 2 (effect protein ligand + tetrazine). Both fragments can penetrate the cell membrane independently and, after entering the body or in vitro, self-assemble into complete TACs through a TCO-tetrazine click chemistry reaction, thereby exerting their target protein degradation effect.
2.2 Photosensitive Linker
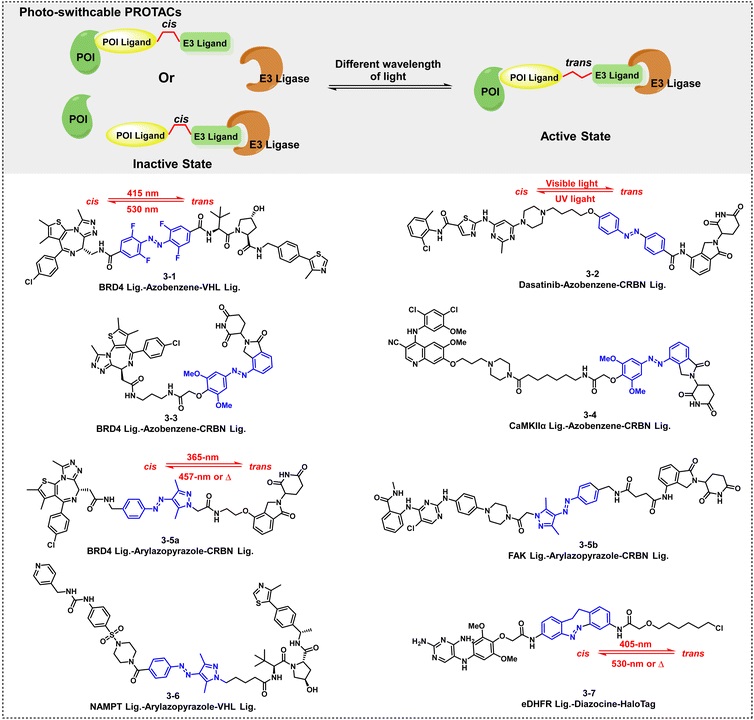
By leveraging the cis-trans isomerism of photoswitching groups (such as azobenzene and arylazopyrazole), the activity of TACs can be precisely controlled. The cis isomer has a distorted conformation and cannot form a ternary complex, thus losing its degradation function; the trans isomer can adapt to the spatial distance between the target protein and the effector protein, activating the degradation function.
A typical example is PHOTAC 3-1, which uses a linker that can repeatedly switch between "degradation-stable" states at 415/530 nm. After illumination, its conformation changes, and the trans isomer allows the target and E3 ligase to bind appropriately, triggering degradation.
2.3 Photocage Linker
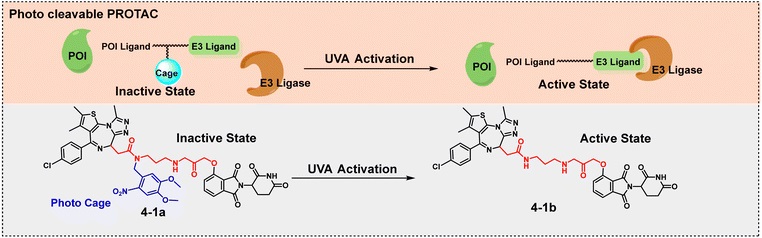
A photocage is introduced into the linker. When not irradiated, TACs are inactive; upon irradiation with light of a specific wavelength, the photocage detaches, releasing the active molecule. For example, under irradiation with a specific wavelength, pc-PROTAC 4-1a detaches its photocage structure, releasing the active degrader 4-1b, which efficiently degrades the BRD4 protein.
2.4 Cleavable Linkers
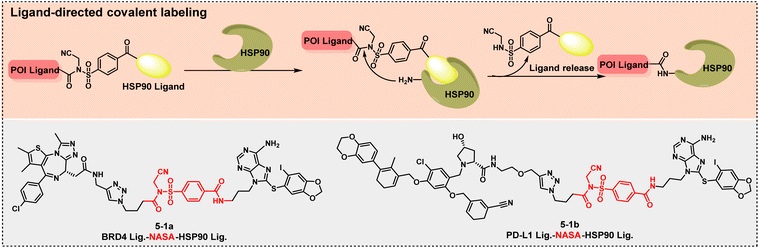
Linkers are cleaved in response to the tumor microenvironment (such as specific enzymes and pH levels), releasing active ingredients. For example, PROTAC 5-1a modifies the HSP90 protein in platelets using NASA technology, forming "degraded platelets (DePLTs)". DePLTs can accumulate at hemorrhagic tumor sites and, upon activation, achieve precise clearance of tumor target proteins through a dual pathway of "ubiquitin-proteasome degradation of intracellular target proteins + lysosomal degradation of extracellular target proteins".
3. Non-Traditional Linkers
Non-traditional linkers break away from the traditional model of "direct chemical linking of target protein ligands and effector protein ligands." Through innovative designs such as split-assembly and nanocarriers, they significantly optimize the cell membrane permeability and tissue targeting of TACs.
3.1 Split-Hybrid Linkers
Target protein ligands and effector protein ligands are split, and each is modified with a self-assembling fragment (such as peptides or lipids). After mixing, they self-assemble into a fully functional degradative agent. Research by Yang et al. showed that modifying target protein ligands such as estrogen receptor α (Era) and cyclin-dependent kinase 4/6 (CDK4/6) with δδRR, while simultaneously modifying E3 ligase ligands such as CRBN and VHL, forms split fragments. After the modules are mixed and self-assembled, they exhibit highly efficient target protein (POI) degradation activity in vitro.
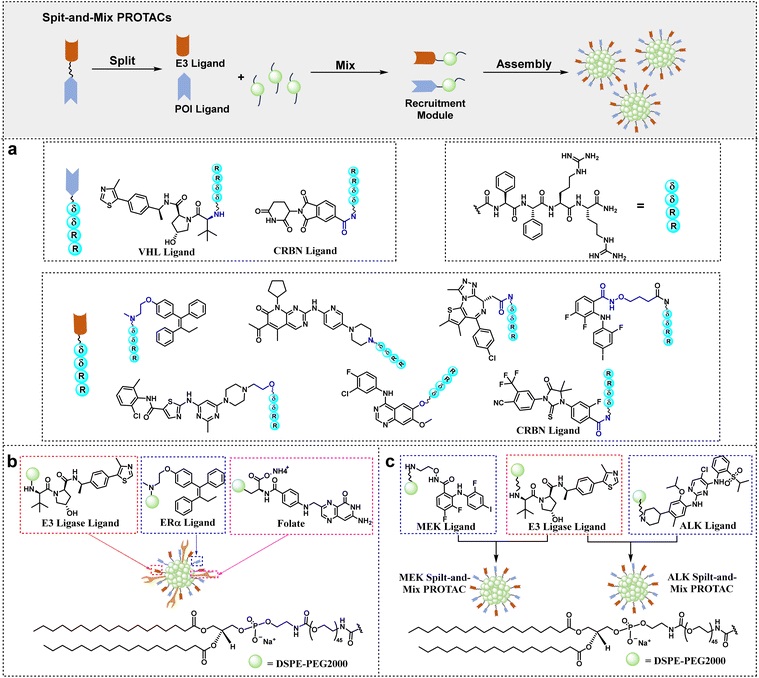
3.2 Self-assembled Nanolinkers
Nanostructures (such as β-sheets and semiconductor polymers) are formed through the self-assembly of peptides or polymers, achieving "long-lasting degradation." Taking Nano-PROTAC (Figure a below) as an example, it forms β-sheet assemblies triggered by GSH (glutathione), which then form assembly-driven monomers via click chemistry, rapidly self-assembling into antiparallel Nano-PROTAC structures. In both in vitro and in vivo experiments, it exhibits highly efficient degradation activity against two representative target proteins, EGFR and AR.
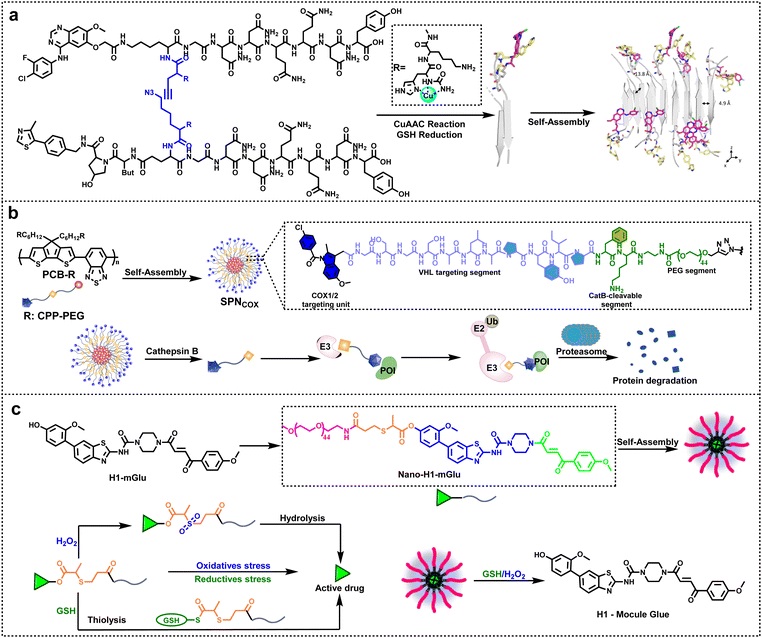
3.3 Gold Nanoparticle (GNP)-based Linkers
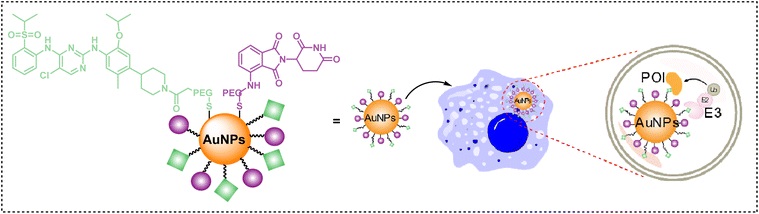
Using gold nanoparticles as the core, PEG-modified target protein ligands (such as ceritinib) and effector protein ligands (such as pomalidomide) are anchored on the surface to form multi-terminal linkers. The synergistic display of multiple ligands can promote the formation of ternary complexes, eliminating the need for complex chemical structure optimization and significantly simplifying the research and development process.
4. Linker Design for Specific TACs
Different TACs have different degradation mechanisms, and their linker design must be precisely matched to the corresponding biological process to maximize degradation efficiency.
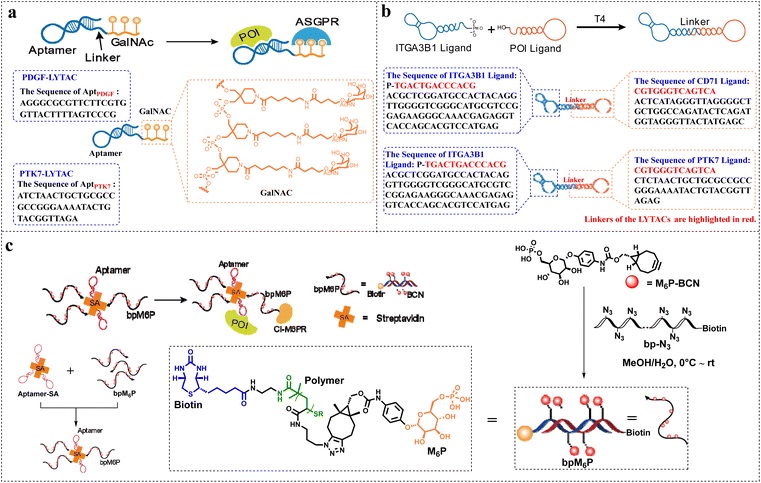
4.1 LYTACs (Lysosome-Targeted Chimeras)
LYTACs deliver target proteins into lysosomes for degradation via endocytosis. The linker needs to be adapted to lysosomal membrane receptors (such as CI-M6PR, ASGPR):
l Polymer-based linker (SPNly)
Composed of semiconductor polymers and IL-4R peptides, it can induce immunogenic cell death after ultrasound activation, while promoting lysosomal degradation of IL-4R, effectively inhibiting tumor recurrence;
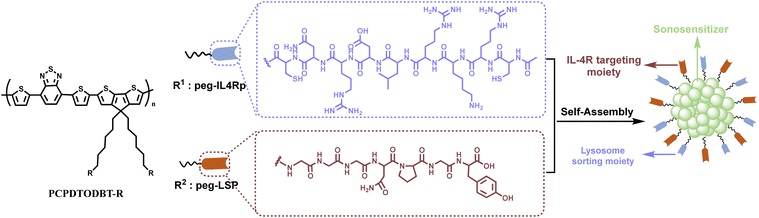
l Aptamer-based linker (Apt-LYTAC)
Targets the hepatocyte membrane protein PTK7 with an aptamer and links it to three GalNAc (N-acetylgalactosamine), significantly improving lysosomal degradation efficiency.
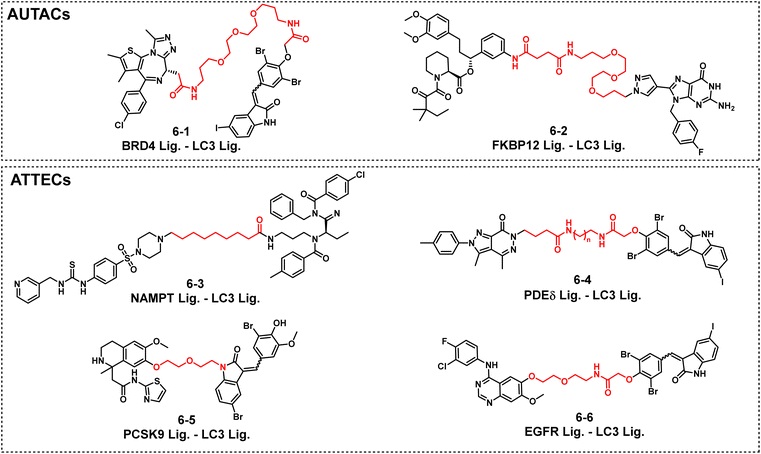
4.2 AUTACs/ATTECs (Autophagy-Lysosome Chimeras)
l AUTACs
Bifunctional molecules composed of a target protein binding ligand, a guanine-based degradation tag, and a linker. The tag guides the target protein into the autophagy degradation pathway. Their linkers are mostly PEG-based (e.g., 6-1), ensuring molecular stability and targeting.
l ATTECs
Act as "molecular glue," directly linking the target protein to the light chain 3 (LC3) on the autophagosome without requiring a degradation tag. Their linkers are mostly flexible alkyl groups (e.g., 6-3), showing significant effects in reducing NAMPT (nicotinamide phosphoribosyltransferase) levels and inhibiting tumor growth.
5. Summary
TACs technology offers a revolutionary strategy for treating intractable diseases such as cancer by degrading "undruggable" targets and overcoming drug resistance. Linker design is a key variable determining its performance. Although there has been innovative progress in linker design, it faces core challenges such as difficulty in balancing multiple properties, lack of "structure-performance" correlation models leading to reliance on experimental screening for research and development, and high cost of synthesizing complex linkers. In the future, with the cross-disciplinary integration of structural biology and other disciplines, TACs linker design is expected to break through bottlenecks and drive more highly effective and low-toxicity TACs drugs into clinical trials.
Read More
https://pubs.rsc.org/en/content/articlelanding/2025/sc/d5sc04859a?sessionid=
