
Gepotidacin
A triazaacenaphthylene bacterial type II topoisomerase inhibitor, a novel antibiotic. It is indicated for the treatment of uncomplicated urinary tract infections in female patients caused by susceptible microorganisms.
Overview
Gepotidacin is a first-in-class triazaacenaphthylene antibacterial agent that simultaneously inhibits bacterial DNA gyrase and topoisomerase IV. This novel dual-targeting mechanism effectively disrupts bacterial DNA replication, offering a promising therapeutic approach for uncomplicated urinary tract infections, particularly in the context of fluoroquinolone resistance.
Synonyms: GSK-944 (maleate); GSK-2140944E; Blujepa; Gepotidacinum; Gepotidacina; Gepotidacin; Buagafuran; GSK-2140944; Gepotidacin(GSK2140944); 3H,8H-2a,5,8a-Triazaacenaphthylene-3,8-dione,2-[[4-[[(3,4-dihydro-2H-pyrano[2,3-c]pyridin-6-yl)methyl]amino]-1-piperidinyl]methyl]-1,2-dihydro-,(2R)-; (R)-2-((4-(((3,4-dihydro-2H-pyrano[2,3-c]pyridinChemicalbook-6-yl)methyl)amino)piperidin-1-yl)methyl)-1,2-dihydro-3H,8H-2a,5,8a-triazaacenaphthylene-3,8-dione; (R)-2-((4-(((3,4-Dihydro-2H-pyrano[2,3-c]pyridin-6-yl)methyl)amino)piperidin-1-yl)methyl)-1,2-dihydro-3H,8H-2a,5,8a-triazaacenaphthylene-3,8-dione,Gepotidacin
EINECS: 202-303-5
Product Categories: APIs; Antibacterials for Systemic Use; Antiinfectives for Systemic Use; BCRP/ABCG2 Substrates; Cytochrome P-450 CYP3A Substrates; Cytochrome P-450 CYP3A4 Substrates; Cytochrome P-450 Substrates; Enzyme Inhibitors; Heterocyclic Compounds, Fused-Ring; MATE 1 Inhibitors; MATE 2 Inhibitors; MATE inhibitors; Naphthalenes; Topoisomerase Inhibitors
Mol File: 1075236-89-3.mol
Physicochemical Properties
Boiling point: 661.9±65.0°C
Density: 1.52±0.1 g/cm3
Storage temp: -20°C
Solubility: DMSO: 7.14 mg/mL (15.92 mM)
Form: Solid
Color: White to Yellow
MSDS Information
Experimental Data
1. Cell Experiment
DMSO : 7.14 mg/mL (15.92 mM; Need ultrasonic)
Preparing Stock Solutions:

Please select an appropriate solvent to prepare the stock solution based on the solubility of the product in different solvents. Once prepared, the solution should be aliquoted and stored to avoid product failure caused by repeated freeze-thaw cycles.
Storage method and duration for stock solutions: -80°C, 2 years; -20°C, 1 year. When stored at -80°C, please use within 2 years; when stored at -20°C, please use within 1 year.
2. Animal Experiment
Please select an appropriate dissolution protocol based on your experimental animals and route of administration. For the following protocols, first prepare a clear stock solution following the in vitro method, then add cosolvents sequentially:
To ensure experimental reliability, the clear stock solution can be appropriately stored according to storage conditions. For in vivo working solutions, it is recommended to prepare them fresh and use on the same day. The percentage indicated before each solvent represents its volume ratio in the final solution. If precipitation occurs during preparation, heating and/or ultrasonication can be used to aid dissolution.
Protocol 1
Add solvents in order: 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% saline
Solubility: ≥ 0.71 mg/mL (1.58 mM); Clear solution
This protocol yields a clear solution of ≥ 0.71 mg/mL (1.58 mM, saturation unknown).
Example for 1 mL working solution: Add 100 μL of 7.1 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix well. Add 50 μL Tween-80 to the mixture and mix thoroughly. Then add 450 μL saline to bring the volume to 1 mL.
Protocol 2
Add solvents in order: 10% DMSO → 90% (20% SBE-β-CD in saline)
Solubility: ≥ 0.71 mg/mL (1.58 mM); Clear solution
This protocol yields a clear solution of ≥ 0.71 mg/mL (1.58 mM, saturation unknown).
Example for 1 mL working solution: Add 100 μL of 7.1 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD in saline and mix well.
Protocol 3
Add solvents in order: 10% DMSO → 90% corn oil
Solubility: ≥ 0.71 mg/mL (1.58 mM); Clear solution
This protocol yields a clear solution of ≥ 0.71 mg/mL (1.58 mM, saturation unknown). This method is not suitable for experiments lasting longer than half a month.
Example for 1 mL working solution: Add 100 μL of 7.1 mg/mL clear DMSO stock solution to 900 μL corn oil and mix well.
Pharmacodynamics
Gepotidacin causes a dose- and concentration-dependent prolongation of the QTc interval. It should be used cautiously (or avoided, where possible) in patients with a history of QTc interval prolongation or those with relevant pre-existing cardiac disease, patients taking antiarrhythmic agents, or other medications that may potentially prolong the QTc interval. Due to an increase in gepotidacin exposure and the subsequent risk of QTc interval prolongation, gepotidacin should also be avoided in patients receiving strong CYP3A4 inhibitors (e.g., itraconazole, ketoconazole), in patients with severe hepatic impairment (Child-Pugh Class C), or in patients with severe renal impairment (eGFR <30 mL/min).
In addition to its bacterial enzyme inhibition, gepotidacin is a reversible inhibitor of human acetylcholinesterase. Patients with medical conditions that may be exacerbated by acetylcholinesterase inhibition should be monitored closely.
Mechanism Of Action
The primary mechanism of action of gepotidacin involves the disruption of bacterial DNA replication through the selective inhibition of two essential bacterial enzymes: DNA gyrase (also known as topoisomerase II) and topoisomerase IV. These enzymes are crucial for bacteria as they manage the topological state of DNA during replication, transcription, and cell division. Gepotidacin exerts its inhibitory effect by binding to these enzymes in a manner that is distinct from other antibiotic classes, including fluoroquinolones - specifically, gepotidacin interacts with the GyrA subunit of bacterial DNA gyrase and the ParC subunit of bacterial topoisomerase IV. Structural studies have revealed that gepotidacin binds midway between the two scissile DNA bonds, within a pocket formed by these subunits.
Similar Products
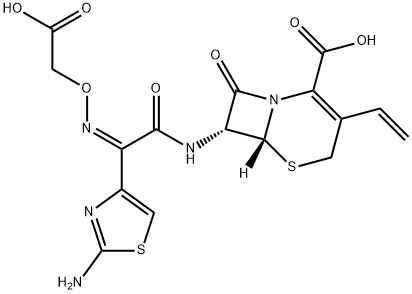
Cefixime
Therapy
: Infectious diseases
Target
: PBPs
CAS
: 79350-37-1
MF
: C16H15N5O7S2
MW
: 453.45
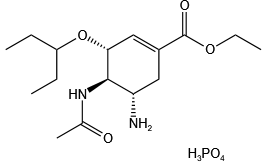
Oseltamivir Phosphate
Therapy
: Infectious diseases
Target
: NA
CAS
: 204255-11-8
MF
: C16H28N2O4•H3PO4
MW
: 410.4
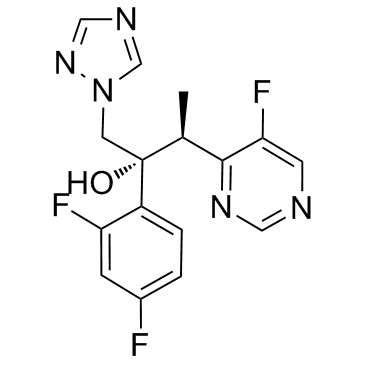
Voriconazole
Therapy
: Infectious diseases
Target
: CYP51A1
CAS
: 137234-62-9
MF
: C16H14F3N5O
MW
: 349.31
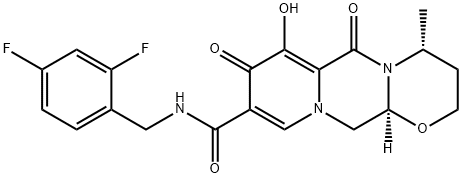
Dolutegravir Hydrochloride
Therapy
: Infectious diseases
Target
: HIV integrase
CAS
: 1051375-16-6
MF
: C20H19F2N3O5
MW
: 419.38
